



Dr. Axel Krieger
Dr. Axel Krieger is an Associate Professor and Carol Croft Linde Faculty Scholar of Mechanical Engineering at Johns Hopkins University. Dr. Krieger’s work focuses on the development of novel tools, imaging, and robot control techniques for medical robotics. Specifically, Dr. Krieger investigates methodologies that (i) increase the smartness and autonomy and (ii) improve image guidance of medical robots to perform previously impossible tasks, improve efficiency, and improve patient outcomes.
Dr. Axel Krieger previously served as Assistant Professor and Head of the Medical Robotics & Equipment Lab at the University of Maryland, College Park. He was also an Assistant Research Professor at the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National, where he led research on robotic tools and laparoscopic devices. Notable projects included the development of the Smart Tissue Autonomous Robot (STAR) and innovative applications of 3D printing for surgical planning and patient-specific implants.
Dr. Krieger holds several licensed patents for biomedical devices. He joined Children’s National after several years of experience in private industry at Sentinelle Medical Inc. and Hologic Inc. His role within these organizations was Product Leader developing devices and software systems from concept to FDA approval and market introduction. Dr. Krieger completed his undergraduate and master’s degrees at the University of Karlsruhe in Germany and his doctorate at Johns Hopkins, where he pioneered an MR-guided prostate biopsy robot used in over 50 patient procedures at three hospitals.
Lab Members

Chunwoo Kim, PhD
Visiting Scholar
Surgical Robots and Systems

Nural Yilmaz, PhD
Postdoctoral Fellow
Surgical Robotics
Control Systems
Deep Learning

Jiawei Ge, PhD
Postdoctoral Fellow
Surgical Robotics

Michael Kam, PhD
Postdoctoral Fellow
Medical Robotics

Laura Connolly, PhD
Postdoctoral Fellow
Surgical Robotics
AI-driven Medical Imaging
Image-guided Interventions

Justin Opfermann, PhD
Postdoctoral Fellow
Robotics

Idris Sunmola, BS
CS PhD Student
AI in Surgical Robotics

Noah Barnes, BS
PhD Candidate
Surgical Robotics

Mariana Smith, BEng
PhD Candidate
Surgical Robotics

Xinhao Chen, BEng
PhD Candidate
Magnetic Actuation System

Michelle Song, BS
PhD Candidate
Surgical Robotics

Ethan Kilmer, BS
CS PhD Student
Surgical Robotics

Juo-Tung Chen, MS
PhD Student
Surgical Robotics
Machine Learning
Research Overview
Our work focuses on both basic research and translational research in the development of novel tools, imaging, and robot control techniques for medical robotics.
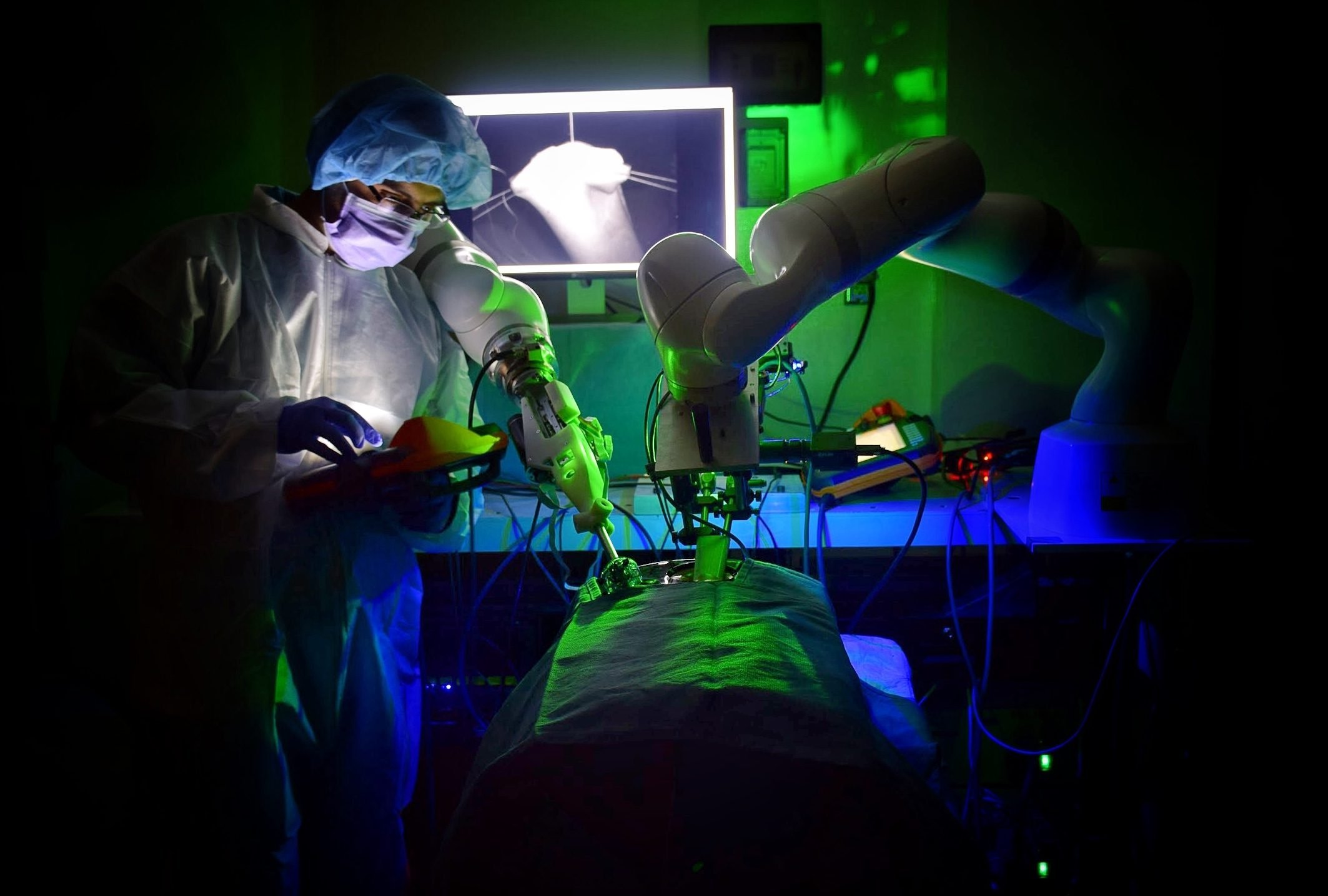
Smart Tissue Autonomous Robot (STAR)
Autonomous surgery holds the promise of providing efficacy, safety, and consistency regardless of individual surgeon skills and experience. The Smart Tissue Autonomous Robot (STAR) represents a groundbreaking robotic platform showcasing surgical automation in image-guided surgery through advanced technologies. The platform seamlessly integrates 2D-3D perception, deformable suture planning, constraint motion control, and deep-learning tissue tracking capabilities, resulting in the achievement of autonomous soft-tissue suturing. STAR recently marked a significant milestone by successfully completing pre-clinical feasibility studies on animal models.
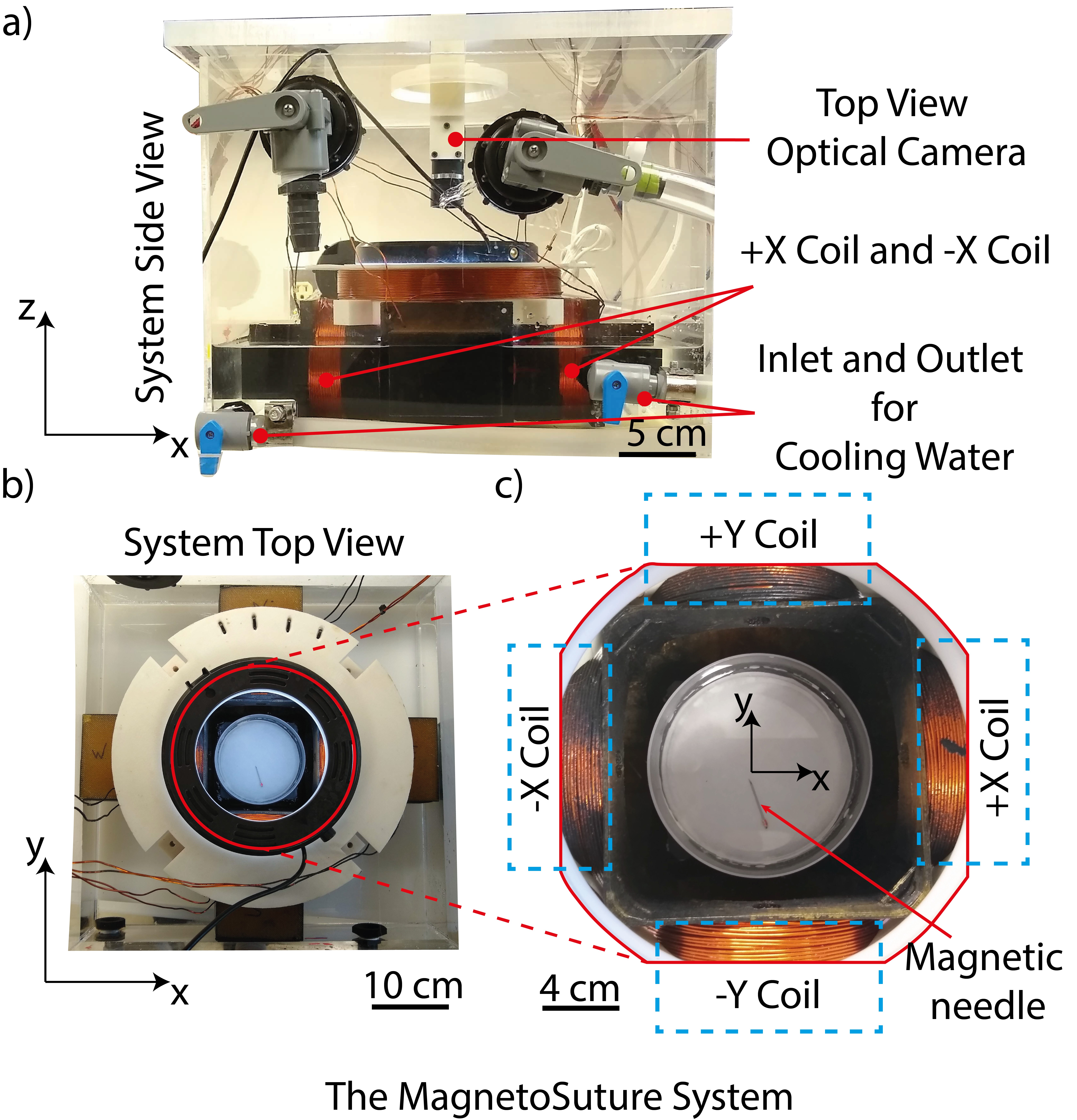
Magnetic Suturing Systems
Magnetic fields can exert forces and torques onto remote magnetic surgical tools that is located inside of the patient’s body, and obviate the physical connections with the standard robotic arm structures. This property of magnetic robotics provides a promising alternative to miniaturize the surgical tools for the next generation of surgical systems, where less tissue trauma and more patient comfort in clinics. As a target medical application, we focus on magnetic suturing, where the needle is magnetic and can be guided to penetrate into the tissue to complete a suturing task. Our research continues towards enhancing the penetration capability and system-level intelligence via merging the digital and physical intelligence.
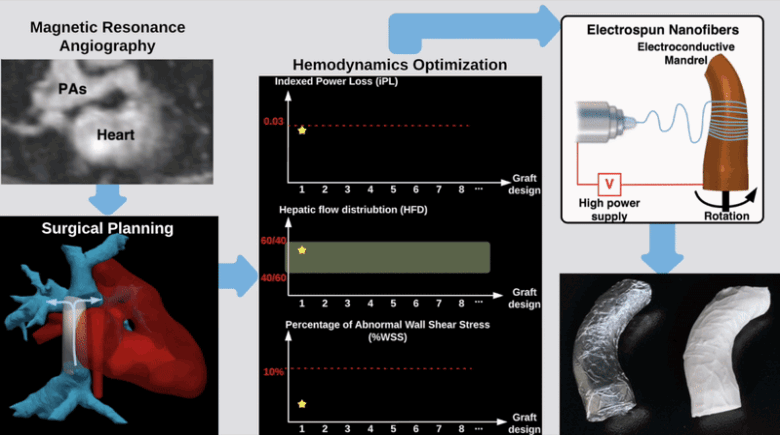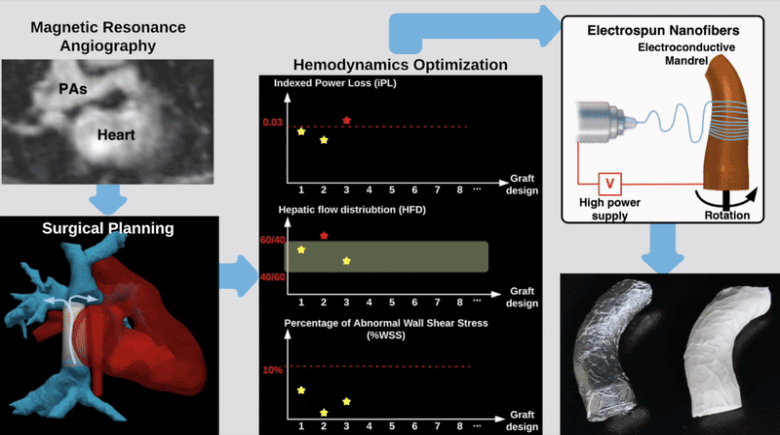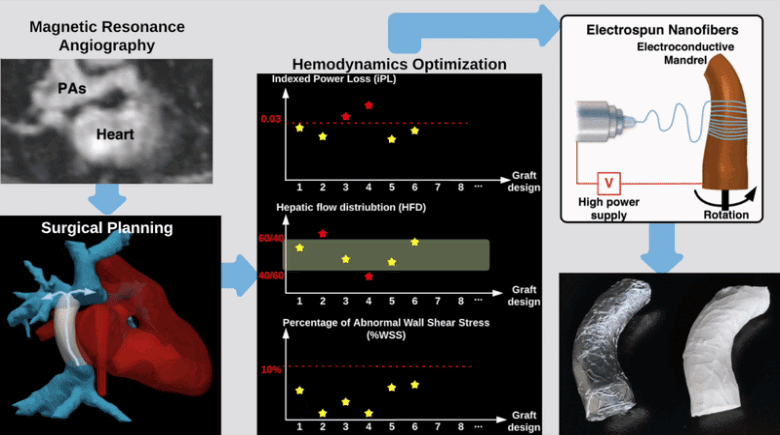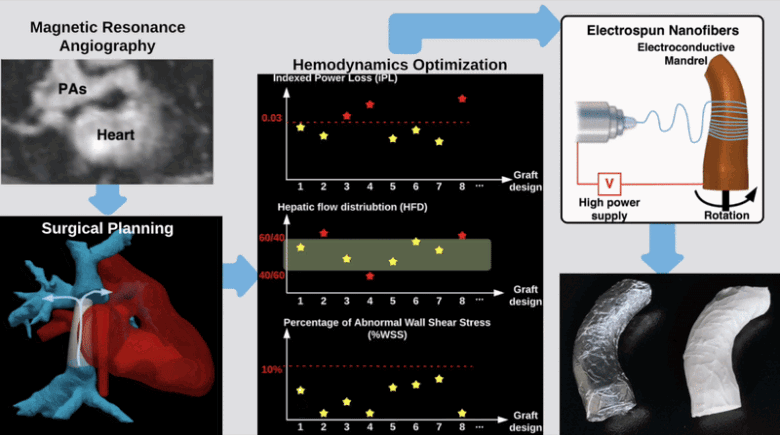
Semi-Automatic Planning and Three-Dimensional Electrospinning of Patient-Specific Grafts for Fontan Surgery
This work aims to develop a semi-automatic tissue engineered vascular graft (TEVG) planning method for designing and 3D-printing hemodynamically optimized Fontan TEVGs. We present a computation framework by parameterizing Fontan grafts to explore patient-specific vascular graft design space and search for optimal designs. We employed nonlinear constrained optimization technique to minimize indexed power loss of Fontan grafts while keeping hepatic flow distribution (HFD) and percentage of abnormal wall shear stress (%WSS) within clinically acceptable thresholds. Our work significantly reduces the collaborative effort and turnaround time between clinicians and engineering teams for designing patient-specific hemodynamically optimized TEVGs.
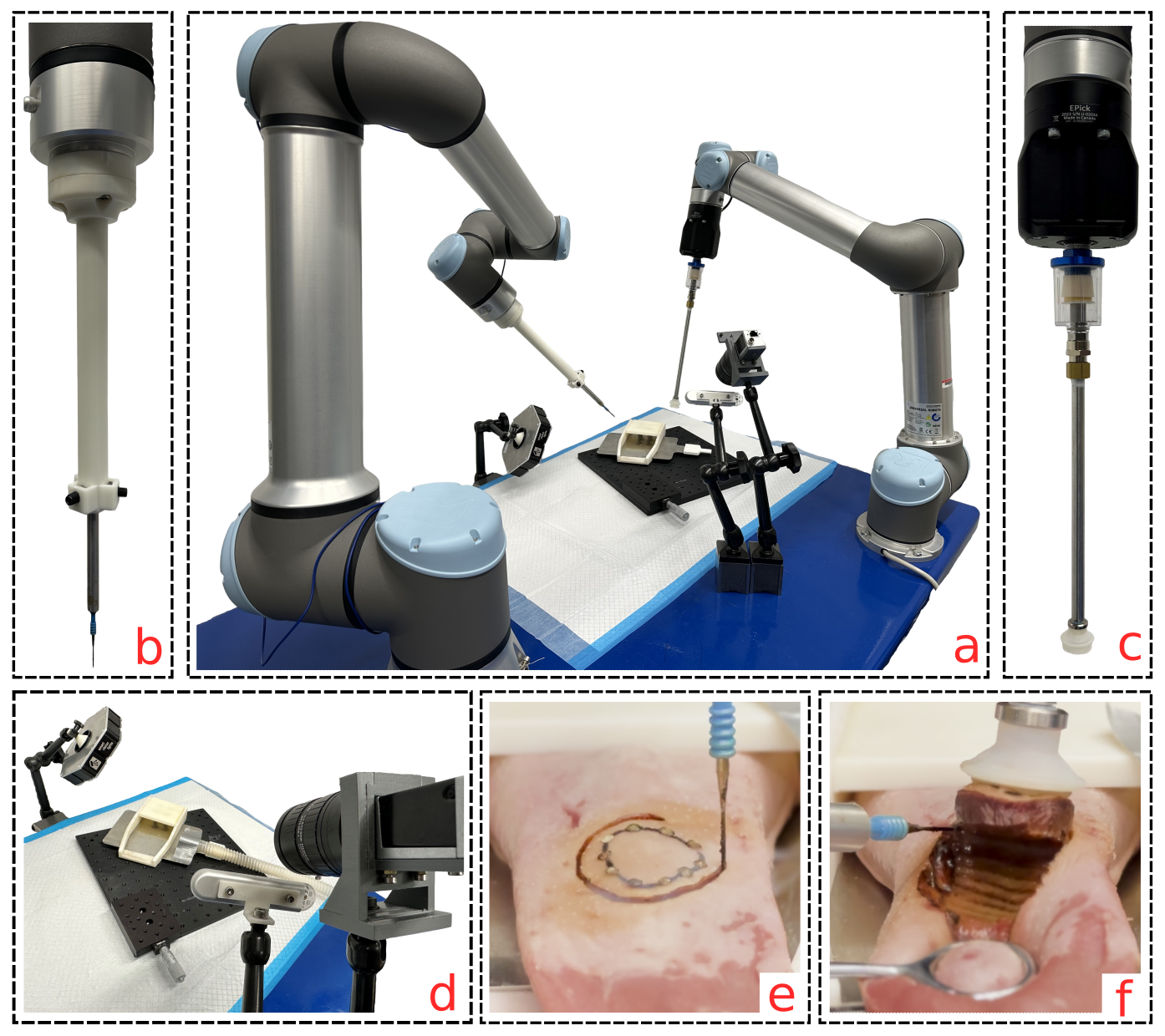
Image-Guided Autonomous Robotic System for Tumor Resection
Tumor resection surgery, a vital cancer treatment, requires the complete removal of tumors and adjacent healthy tissues, demanding high surgical precision for optimal oncologic outcomes. We developed the autonomous system for tumor resection (ASTR), a pioneering dual-arm, vision-guided robotic system tailored for this purpose. Demonstrated in a glossectomy-mimicking surgical setup using porcine tongue samples, ASTR’s successful autonomous performance yielded no positive margins, showcasing precision and consistency that rival or even surpass manual resections by experienced otolaryngologist.
Research is fun, but it’s even better with fun activities!
Lab Gallery


Latest News from Our Lab
Two Papers to be Presented at IROS 2024
Two papers have been accepted and will be presented IEEE/RSJ International Conference on the Intelligent Robots and Systems (IROS) in Abu Dhabi, United Arab Emirates, October 14-18, 2024. Tracking Tumors[…]
Read moreCongrats Lidia’s research featured on the Hopkins Engineering News
When Minutes Matter: Robot Tackles Trauma’s Silent Killer
Read moreContact us
Please reach out if have any questions about the research or opportunities.
